I have created this outline as per my experience during the assessments (ISO 17025 Accreditation). It seems that after reading the manual, you easily forget the next requirements to do. My first time reading it (ISO 17025 Standard) is very overwhelming.
#Iso 17025 2017 management review template iso
Laboratory accreditation to iso iec 17025.One of the most challenging parts during establishing a calibration or testing lab is to start the documentation requirements, even if there is an established guide (ISO 17025) that we can read and follow. Section 8 - Management System Requirements: Options, Management System Documentation, Control of Management System Documentation, Control of Records, Risks and Opportunities, Improvements, Corrective Actions, Internal Audit, Management ReviewsĬanadian Association for Laboratory Accreditation Section 7 - Process Requirements: Review of requests, tenders and contracts, Method Selection, Sampling, Handling of Customer Items, Technical Records, Evaluation of Measurement Uncertainty, Validity of Results, Reporting, Complaints, Nonconforming work, Control of Data and Information Management (NEW) Section 6 - Resource Requirements: General, Personnel, Facility and Environment, Equipment, Metrological Traceability, External Products and Services Section 5 - Structural Requirements: Legal Entity, Management, Range of Laboratory Activities (NEW), Conformity, Availability and Authority of Resources, Personnel and Integrity

Section 4 - General Requirements: Impartiality and Confidentiality The new requirement sections are as follows: There are now 5 requirements sections instead of the 2 requirements sections (section 4 and 5) from the 2005 version of the standard. The ISO 17025 standard was re-issued in 2017 as a replacement to the 2005 version. IEC: International Electrotechnical Commissionġ7025: Unique standard number assigned by ISO

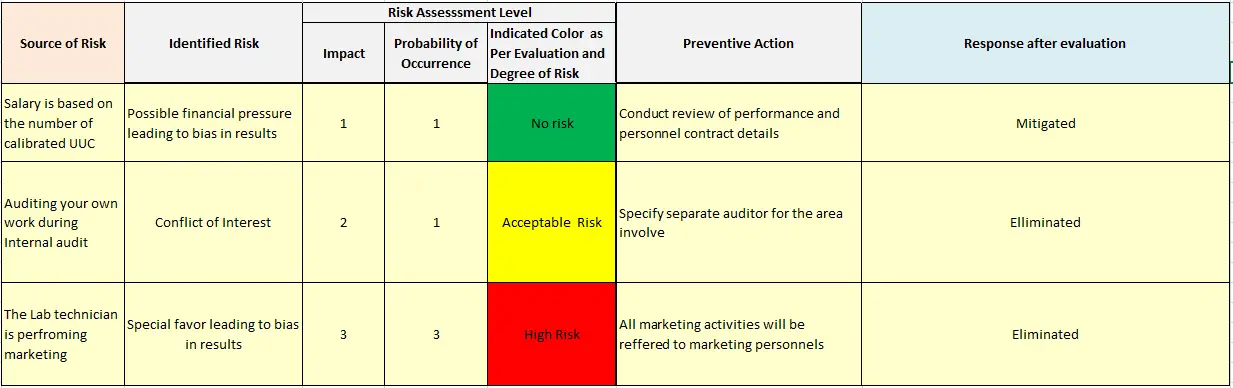
ISO: International Organization for Standardization ISO/IEC 17025:2017 - General requirement for the competence of testing and calibration laboratories The laboratory needs a plan to address risks, however, there is no requirement for a formal risk management system or documented risk management process.Ĭontents of the ISO/IEC 17025:2017 International Standard The ISO/IEC 17025 standard can be obtained through ISO at the following link: This can be accomplished with policies and procedures if the lab prefers or by evidence of records such as training, internal audit, etc. For example, ISO/IEC 17025:2005 stated "the laboratory shall have policies and procedures to ensure protection of confidential information" and ISO/IEC 17025:2017 states "the laboratory shall ensure the protection of confidential information.". There is more latitude on how laboratories can implement the standard. Risk based thinking applied in this version enables some reduction in prescriptive requirements and their replacement by performance based requirements. The ISO/IEC 17025 international standard was revised in 2017.Īddition of processes rather than procedures, risk based thinking and management, identifying opportunities in business, quality and customer satisfaction, measured performance, prevention of negative impacts, effective improvements, separated technical and Management System Records.
